The ‘New’ Kid on the Block: mRNA vaccines and why they might just be here to stay

The development of vaccination programmes around the globe has seen pivotal positive effects of vaccines upon public health, raising the discussion that vaccines are one of humanity’s greatest inventions. It all started with Edward Jenner, who we can all thank for developing the world’s first successful vaccine against smallpox in the late 1700s. From that point, not only has smallpox been considered “eradicated” by the World Health Organisation but methods and scientific know-how of vaccines have shifted as well.
Are messenger RNA (mRNA) vaccines new?
The short answer is no. Although mRNA vaccines are newly licensed introductions to prevent coronavirus infections. Despite this new authorisation, research has been firmly established for decades, with mRNA clinical trials involving diseases such as influenza, rabies, zika to cancer have been studied.1
mRNA vaccine research really kicked off in the late 1980s2 prior to its authorisation in preventing COVID-19 infection.
Scientists, for over two decades, have been analysing the use of mRNA as a possible therapeutic option for some malignancies. Recent clinical trials for melanoma have yielded positive results where 8 out of 13 patients saw reduced risk associated with metastasis lesion formation, potentially opening doors for enhancing clinical outcomes of cancer.3
So, what exactly is mRNA?
To understand mRNA vaccines, we must first understand the role mRNA has in basic human biology. mRNA plays a core role in protein synthesis and in turn coordinating critical biological function, but how? To answer this, let’s delve into the inner workings of a human cell.
Zooming into the primary location of the genome within the cell, we find ourselves situated within the nucleus. Here, DNA contains genetic information required for protein production (fundamental in sustaining life). Considering DNA as the brains here, due to its function, it cannot produce proteins by itself so it ‘outsources’ this to mRNA instead. mRNA copies a version of the DNA gene containing instructions required to produce a protein of interest. This mRNA molecule acts as a messenger, leaving from the nucleus into the cell cytoplasm, delivering this information to ribosomes. Here, within the intracellular space, ribosomes ‘read’ along the mRNA molecule assembling the desired protein.4
A key point is that scientists learned decades ago that, if you supply a cell with external mRNA, the cell will use it to produce whatever protein is encoded by those instructions.5
How do mRNA Vaccines work?
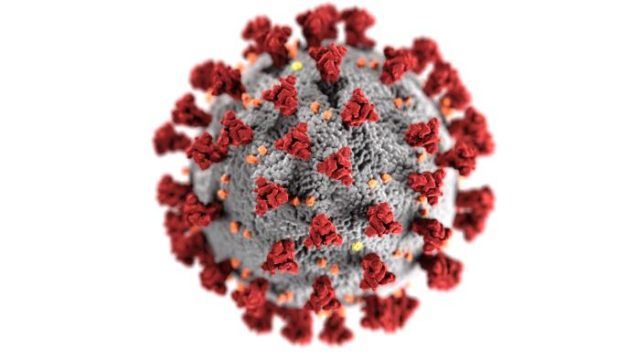
Having a glance above of the red area of the viral membrane of a severe acute respiratory syndrome coronavirus 2 (SARs-Cov-2). This is the spike protein. Its function is to bind to host cells, allowing entry and following viral replication within our cells results in respiratory illness and transmission.
mRNA vaccines utilise the mRNA strand which codes for the specific protein, in this case, a spike protein from SARS-Cov-2. Ergo, the vaccine provides your own cells with the mRNA necessary to synthesise the spike protein through methods discussed earlier. After being synthesised, these proteins act as a “non-self” beacon to your immune system to signal the creation of antibodies. The main takeaway here is that mRNA vaccines produce a specific protein from a specific virus, driving a specific immune response.6
What separates mRNA from the crowd?
It’s important to state here that all vaccines developed, irrespective of type are ALL key in reducing infectious diseases. The real trick is the use of all vaccine types to achieve the shared goal of reducing infectious disease in a population.
- Suitability for the immunocompromised?
To be immunocompromised is in its name. This refers to people whose immune systems are debilidated as a result of various factors: such as undergoing chemotherapy or bone marrow transplants, to infections such as HIV. The mRNA vaccine does not contain the live attenuated strain of SARS-CoV-2 so this could in fact make it suitable for those who are immunosuppressed, despite being less effective.7
- Quick production
Unlike the conventional vaccine types, although good as they are, mRNA has the potential for faster manufacturing times, giving them an edge in handling emerging infectious diseases that require rapid response. Usually, acquiring an experimental traditional vaccine takes months due to time taken to accumulate the virus, ensuring large amounts are cultured and properly handled, then onto distribution.8
On the contrary, mRNA production avoids extra steps, which would otherwise extend the production timeline of these vaccines as utilised by traditional vaccine technologies. Regardless of this, production of mRNA vaccines is laborious, requiring multiple complex stages with quality assurance checks compensating for more than half of the production time process! To produce a batch of Pfizer-BioNTech vaccine under established procedures takes 110 days from start to finish with this number said to be reduced to just 60!9
- Increased biosafety
Notably, viruses such as SARS-CoV-2 require a level 3 biosafety facility, which complicates procedures further acting as a biosafety threat. However, for mRNA vaccines, only small quantifiable amounts are required, for gene sequencing and testing efficacy of the vaccine upon viruses.10
Current distribution difficulty
If you could mention the biggest downside to mRNA despite its great potential, distribution capabilities would be its Achilles heel. As a molecule, mRNA is fragile and can only be maintained optimally at specific cold temperatures. Any degree of fluctuations from hot or cold can denature the molecule, rendering the vaccine benefits to a minimum, whilst arising difficulties for countries who may not be financially privileged. The Pfizer-BioNTech vaccine needs to be stored at lows between -60°C – -80°C.11
However researchers alluded that due to time pressures to get the vaccine safely and quickly authorised, there was not enough time to optimise the storage at higher temperatures. Moderna’s mRNA vaccine on the other hand can be stored at -20°C. As research jabs forward, increasing thermal stability of mRNA vaccines may reduce distribution difficulties, hopefully widening global outreach of these vaccines.12
The End and also the beginning…
So, there’s the gist of mRNA vaccines, and as for what comes next is still to be explored. One question to ask is are we at the tip of the iceberg in curbing infectious disease using mRNA technology? Of course, this is a question that requires more research, time, and investment. However, going by the clinical trial promise and the successful production we have seen against COVID-19, mRNA is convincing the world and companies to invest in this type of research and development as we look towards the future.
This article was specialist edited by Giuditta De Lorenzo and copy-edited by Richard Murchie
References
- https://www.nature.com/articles/nrd.2017.243
- https://www.pnas.org/content/86/16/6077.short
- https://www.nature.com/articles/nature23003
- https://www.genome.gov/genetics-glossary/messenger-rna
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7076378/
- https://www.medicalnewstoday.com/articles/how-do-mrna-vaccines-work#What-is-diffrent-about-mRNA-vaccines?
- https://www.immunology.org/news/bsi-statement-covid-19-vaccines-for-patients-immunocompromised-immunosuppressed
- https://biontech.de/covid-19-portal/mrna-vaccines
- https://eu.usatoday.com/in-depth/news/health/2021/02/07/how-covid-vaccine-made-step-step-journey-pfizer-dose/4371693001/
- https://www.bulblaboratories.com/coronavirus-handling-biosafety-levels
- https://www.cdc.gov/vaccines/covid-19/info-by-product/pfizer/index.html
- https://www.thelancet.com/journals/eclinm/article/PIIS2589-5370(21)00026-2/fulltext










