Stem Cell Etiquette
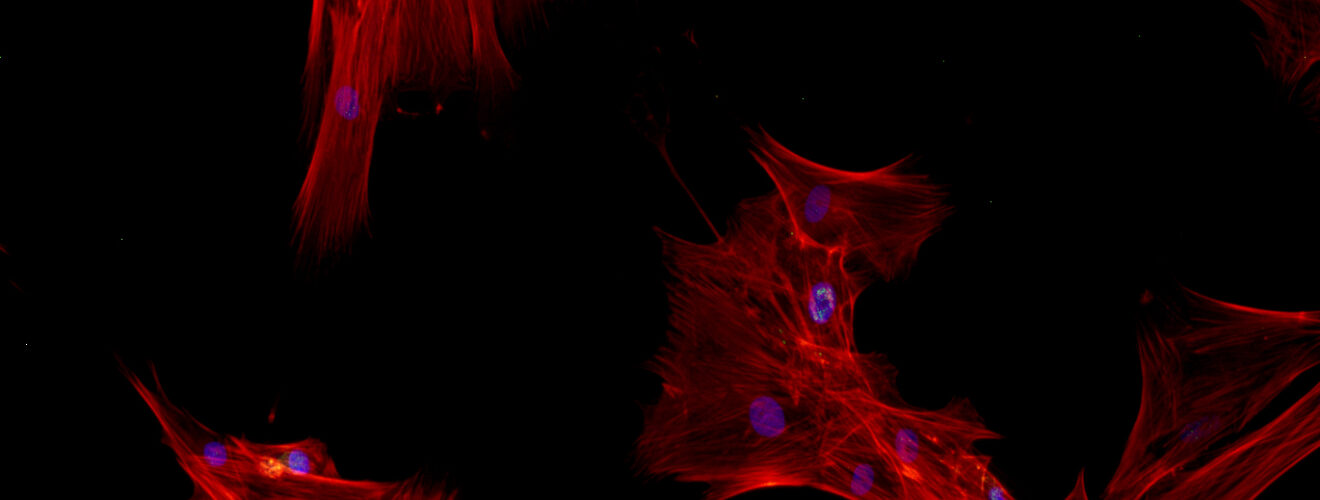
An eukaryotic cell, complex little life machine that it is, goes through a whole lot over its lifespan: it grows, divides, communicates with other cells and harbours a myriad of signals and processes in the process. If it happens to be a stem cell, it also has to tackle the monumental task of differentiation: from its privileged state, with the potential to produce different cell types, it progresses to a committed “career” or, more scientifically put, a lineage.
Differentiation in the human body is pretty much a given, for example during the development of an embryo or, in adult life, for the generation of blood cells from adult stem cells present in the bone marrow. Out of the body and into the lab however, a lot of research goes into how to get stem cells to behave the way we want them to; this can be done by stimulating the cells chemically or physically. Since cells in the lab are mostly grown on plastic surfaces inside tiny flasks, scientists have tried altering these surfaces by attaching chemical molecules to them, changing their stiffness or adding patterns such as pits and pillars to them, and then simply seeing what happens to the cells.
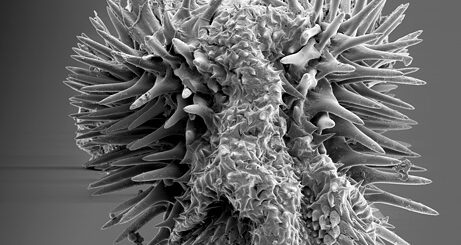
For example, in our own neck of the woods, scientists at the University of Glasgow and the University of Strathclyde have been experimenting with nanotopography, adding features only a few hundred nanometres in size to surfaces and culturing particular types of stem cells on them – specifically mesenchymal stem cells, which can differentiate into bone, fat and cartilage. Interestingly, they found that cells behave differently on different topographies, sometimes turning more readily into bone cells while other times promoting the generation of more stem cells for longer periods of time 1.
These findings show a lot of promise: mesenchymal stem cells can be used for various regenerative therapies but are notoriously difficult to obtain from patients and to store for future use. Having a surface which actively promotes their “stemness” would thus be an invaluable medical advantage. The ability to direct stem cells to form bone tissue would also be welcome in the field of orthopedics for instance, leading to implants with patterns which stimulate bone formation and integrate better in the body. This new and exciting field is only just starting to gain the recognition it deserves, so we may expect a lot more good news in the future.
Edited by Sarah Spence