Defusing the neurology time bomb- A new gene therapy for Parkinson’s Disease
Parkinson’s Disease (PD) – familiar to many because of the philanthropic efforts of the Michael J Fox Foundation – is a pernicious neurological disease that affects 1% of people over 60 1. The financial burden on the NHS is estimated to be between £449 million and a staggering £3.3 billion per year 2, yet the dominant issue for many patients is worrying about being a burden on their families and the associated emotional consequences. The failure to adequately treat PD coupled with the effects of an ageing population has been described by Parkinson’s UK as a “neurology time bomb”3. They predict that the number of patients will increase by 28% by 2020, while incidences of cancer and heart disease will decrease in the same period. It’s of paramount importance that more effective treatments for PD should be developed if we don’t wish to incur the increasing economic and human cost of the disease.
At the molecular level, PD is the disruption of key neural pathways by the progressive death of neurons that produce dopamine, a chemical that allows communication between neurons. By mechanisms, which are poorly understood, large blobs of disorganised protein known as Lewy Bodies develop inside brain cells. This process physically disrupts the important biological processes that are essential for life. As neurons are killed off by Lewy bodies, connections are severed between parts of the brain – especially the nigrostriatal connection between the substantia nigra and the striatum caudate, regions of the brain involved in coordinating addiction, reward, movement and motivation. An individual is said to have PD if about 70% of the cells that form this connection have been killed, but the disease is progressive and does not stop at 70%. PD will continue to cause further damage until the end of the patient’s life, while the symptoms gradually worsen.
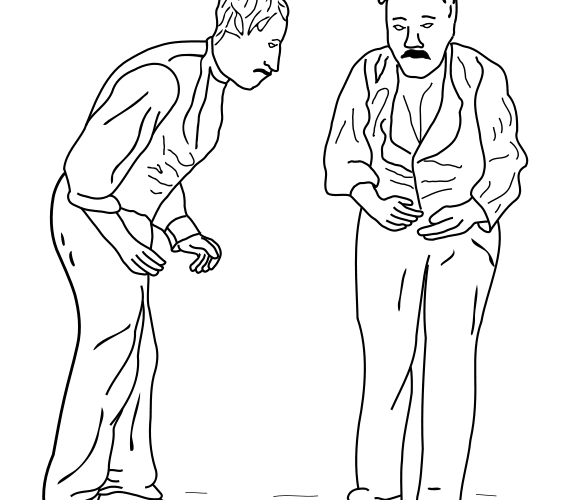
The symptoms of PD have been documented by physicians for hundreds of years: the sketch4 shown below is of a PD patient who has been suffering from mobility problems – a hallmark of the condition. The destruction of the nigrostriatal pathway prevents proper coordination of motor function. Many of the associated symptoms can be seen in the way a patient walks: with a lack of unconscious movement such as arm-swinging, shuffling instead of walking, slow movement, or involuntary movement like shaking.
The severity, onset and type of symptoms vary wildly from patient to patient. Some will experience problems with vision or hearing; others will be unable to speak or sleep. These symptoms usually appear in the early stages of the disease with more severe psychiatric disorders, such as dementia, developing later. Patients in the later stages of the disease often require around-the-clock care and supervision as they are unable to carry out simple tasks like preparing meals or, indeed, being able to swallow food at all 5.
Typically, patients are treated with the drug L-DOPA, which is converted to dopamine in the brain to alleviate some of the symptoms. It is not a cure, however, because L-DOPA becomes less effective as PD progresses and more neurons are killed but it does provide some relief for early-stage symptoms. L-DOPA treatment is effective for about 5-10 years during the earliest stages of PD. As with all therapies, there are also some side effects: the drug causes hugely fluctuating levels of dopamine which results in alternating involuntary muscle movements and a loss of voluntary movement (dyskinesia and akinesia). It has been suggested that this fluctuation with L-DOPA and similar drugs can cause psychiatric disorders such as pathological gambling, psychotic behaviour, sleep disorders and extreme levels of anxiety 6.
University of Oxford spin-out company, Oxford Biomedica promises that their new therapy, ProSavin, can reverse PD after just one administration with none of the side effects of L-DOPA 7. ProSavin is a gene therapy-based treatment that uses a modified version of the horse equivalent of HIV, equine lentivirus, to deliver a set of three genes into the brains of PD patients. The genes are inserted into the DNA of neurons that don’t normally produce dopamine so that they can be reprogrammed into making dopamine to compensate for the deaths of the neurons that can. This therapy is administered surgically and is designed to last years before patients require another dose – this is particularly useful to those with PD-associated memory loss symptoms who could neglect to take their daily doses of L-DOPA 8.
The therapeutic effect was first demonstrated in a study carried out on Macaque monkeys with PD induced by the neurotoxin MPP+, which disrupts the mitochondria within dopamine-producing neurons, thus killing them. ProSavin was found to alleviate some of the motor control symptoms of PD and did not produce the side effects that were seen in the Macaque population treated with L-DOPA. The results of these experiments were encouraging and so ProSavin was moved into what is referred to as the “first in-man” trial or Stage 1 clinical trial, where the safety and the basic therapeutic effect of a treatment are confirmed in a small group of human patients. Fifteen PD patients who were experiencing side effects from their L-DOPA medication were given ProSavin and were interviewed regularly for up to fourty-eight months to check for any side effects. Some cases of a lack of motor control were recorded in patients taking the highest doses of L-DOPA, yet, overall, their conditions did improve with ProSavin and the treatment was deemed to be effective and safe enough for further study 9.
The authors of the study recommend that ProSavin be tested in a more rigorous randomised double-blind placebo-controlled trial with more participants and a longer follow-up period to ensure that it is safe and effective. Patient recruitment is ongoing, and the trial period is likely to last between ten and fifteen years 10.
ProSavin has been promoted as “the cure” for PD and the results of the first trials suggest it is safe in the short term and has a measurable therapeutic effect. More research is required to be sure of the long-term effects, but medical science might be that bit closer to defusing this neurology time bomb.
Defusing the neurology time bomb- A new gene therapy for Parkinson’s Disease by Euan Wilson was specialist edited by Tina Goldie and copy edited by Charlie Stamenova.
References
- de Lau L; Breteler M, “Epidemiology of Parkinson’s disease”, The Lancet Neurology. 2006.
- Findley LJ, “The economic impact of Parkinson’s disease”, Parkinsonism & Related Disorders. 2007.
- BBC article on the matter.
- Gowers R, “A manual of the diseases of the nervous system”, 1886, J & A Churchill
- Weerkamp NJ et al, “Nonmotor symptoms in nursing home residents with Parkinson’s disease: prevalence and effect on quality of life”, J Am Geriatr Soc. 2013
- Merims and Giliad, “Dopamine dysregulation syndrome, addiction and behavioral changes in Parkinson’s disease”, Parkinsonism & Related Disorders. 2008.
- Read more about it here.
- Jarraya B et al, “Dopamine Gene Therapy for Parkinson’s Disease in a Nonhuman Primate Without Associated Dyskinesia”, Sci Transl Med. 2009.
- Palfi S et al, “Long-term safety and tolerability of ProSavin, a lentiviral vector-based gene therapy for Parkinson’s disease: a dose escalation, open-label, phase 1/2 trial”, The Lancet. 2014.
- Read more about the trials here.