Fighting cancer with junk?

Approximately one in every six people worldwide dies each year from cancer; it is one of the leading causes of death globally, second only to cardiovascular disease[1]. In the UK alone, around 100 people are newly diagnosed with cancer every day, and predictions indicate that this figure will rise by around 2% in the next 15 years[2].
Humans are not the only species to develop cancer. In fact, cancers are common throughout the animal kingdom. But did you know that some animals are actually cancer resistant! From large mammals like elephants to comparatively tiny bats, cancer resistant creatures exist[3]. Bizarrely, included among this list are several species of rodents collectively called mole rats, which rarely develop cancer unlike other members of the rodent family. Rats and mice, for example, can develop the disease rapidly. Studies suggest that mole rats may have evolved different, species-specific methods to prevent cancer establishing and growing. Finding out how they do this may be a key factor in defeating cancer in human cells. Here, we investigate cancer as a disease and how one species of mole rat, the blind mole rat, has evolved to tackle it.
What is cancer?
The word ‘cancer’ describes a group of complex diseases with a range of symptoms. Cancer can affect almost any part of the human body, though some regions are more susceptible than others. In the UK, lung and bowel cancer (accounting for 21% and 10% of cancer deaths respectively) are some of the most common types, whilst cancers such as those of the stomach are rarer (claiming around 3 % of the lives taken by cancer each year)[4]. But how do we get cancer?
Almost all human cells contain deoxyribonucleic acid (DNA) located inside a specialised compartment called the nucleus. The information encoded in human DNA, can be accessed by the cells. Just like the books on a library shelf, DNA is arranged on structures called chromosomes (as genes), which store genetic information in a neat and compact way. Genes contain the instructions (i.e., specific DNA sequences) the cell needs to make complex molecules called proteins. Specialised proteins in the cell read and translate the DNA information to make other proteins. Some of these specialised proteins are known as transcription factors and help a cell to turn ‘on’ and ‘off’ certain genes to control which proteins are made and when. The cocktail of proteins present in a cell at any given moment helps determine the type of cell it is, its current state of activity, and the functions it carries out. For example, the hair on our heads is mostly made of a protein called Keratin, made by a type of cell in your body called a Keratinocyte cell[5]. Despite containing the same DNA as in our Keratinocyte cells, the cells we find in our stomachs, which make hydrochloric acid to breakdown our food (Parietal cells)[6], are not responsible for making the Keratin we find in our hair. The two different kinds of cells can regulate how the information in their DNA is used and specialise to perform different functions in the body.
Cancers begin when cells in the body divide abnormally and too quickly. In many cases, this causes a lump of abnormal cells to form (a tumour), and sometimes go on to escape from their original location, travel around the body, and invade other areas: in other words, they cause the cancer to spread (metastases). When the cells divide (i.e., double in number), the entire DNA library is copied carefully to prevent mistakes, so that each new daughter cell inherits one complete and accurate copy. However, mistakes can cause a region (or regions) of DNA to disappear or become damaged. In some cases, mistakes can even occur when a cell performs too many divisions. Collectively, these mistakes are known as mutations. Mutations happen naturally within a cell and can be useful if their changes allow cells to adapt their DNA to changes in their environment. Some mutations are, however, associated with cancer.
Cancer-associated mutations can be induced through exposure to cancer-causing chemicals (e.g., in smokers) or a family history of cancer. When these mutations prevent cells from understanding the information in their genes, problems can occur. For example, if the cell makes too much or too little of a protein, it might lose its ability to start or stop copying its DNA before the cell is ready. Mutations may also result in different versions of a protein being produced, causing its original function to change and its behaviour within the cell to become abnormal[7]. At the simplest level, then, cancer could be described as a disease of the DNA. Of course, the reality is far more complex, and often multifactorial.
Rodents with superpowers: the blind mole rat
Looking at a blind mole rat (species Spalax), one might easily doubt that they are natural cancer-fighting machines. These small, brown/black rodents live around South-eastern Europe and North Africa, burrowing through the dirt using their large front teeth[8]. The lifestyle of these furry creatures is challenging — they live underground in burrows — yet, it is a lifestyle to which they are well adapted. They need very little oxygen, tolerating levels as low as 3% for up to 14 hours without showing signs of illness[9]. For comparison, humans need around 20% oxygen in the air to survive; where oxygen falls between 10-14%, human bodies begin to shut down and at lower levels cease to function[10]. Surprisingly, these unassuming, furry soil-dwellers never develop cancer spontaneously, nor are they at increased risk of developing it as they age, unlike humans. In fact, the lifespan of these odd little creatures is reported to be around 20 years; only around three times less than that of the average human being (72.6 years)[11]. Mole rats also live almost ten times longer than their fellow rodents, mice and rats, whose life expectancy is around two years. But why don’t they get cancer? To understand why, we need to take a small step back and look at our DNA.
Human cells have around 3 billion base pairs’ worth of DNA each, but little is known about what roughly 98% of it does[12]. For a long time, the ‘extra’ bits of DNA were dismissed as junk, but the more we study our ‘junk DNA’ (called non-coding DNA), the more we realise its importance. Some ‘junk DNA’ is made up of DNA sequences called transposable elements (TEs). These are pieces of DNA which can move around; and are sometimes called jumping genes. Close to 50% of our DNA sequences originate from TEs. One group is called the DNA transposons, but they are considered inactive. The second group, retrotransposons (RTs), which come from pieces of copied RNA jump from one region to another. Evidence suggests these sequences of DNA may be leftover pieces of ancient viruses, and given their apparent inutility, it is unclear why we keep them in our DNA sequence. In fact, our cells actively try and stop RTs from moving around and causing problems; in a process known as methylation, specialised enzymes (methyltransferases) add methyl groups to the RTs to inactivate them[13]. Sometimes, however, these safeguards fail, and RTs become activated; this can happen as we get older and may explain some of the aging we see naturally in our brains[14].
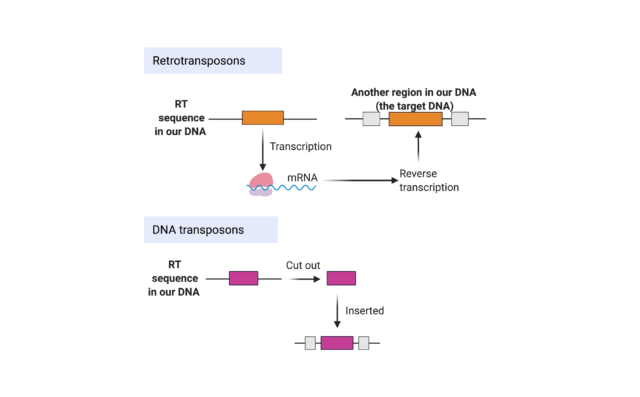
Blind mole rats fight cancer with ‘junk’
When cells of the blind mole rat start to replicate faster and become more like cancer cells, they begin producing a biological chemical called Interferon beta (IFN-β). IFN-β is a small protein, called a cytokine, which can be made by some cells in the immune system to coordinate parts of the immune response. For example, IFN-β can be produced by macrophages, immune cells which attack and consume other cells. This increase in IFN-β causes the abnormal cells to die instead of becoming cancerous. However, what causes the cells to make more IFN-β was, until recently, somewhat mysterious. A closer look at what was happening in the cells of these unusual rodents uncovered a key plot twist; RTs were causing the cells to make IFN-β[15]. The blind mole rats were using some of their ‘junk DNA’ to attack and kill cancer before they developed further. When these RTs became more active, researchers also noticed that there were reduced levels of methylation in the precancerous cells (note that methylation is ordinarily used to keep RTs silent). Remarkably, either by forcing cells to reduce the level of methylation or by giving them more RTs, scientists have been able to stop the growth of human cancer cells in the lab. Whereas, when blind mole rat cells were given drugs used to prevent the RTs from being produced (i.e., antiviral drugs), the precancerous cells were not killed.
We might then ask, given the remarkable findings in blind mole rats, whether we also have something in our own, human ‘junk DNA’ with similar therapeutic power. Though much more research is needed to really understand how the mole rats keep their RTs in check whilst activating them to kill cancer, these results could represent a key breakthrough in how we think about cancer and the potential that could be hidden away in our own DNA.
Edited by Holly Leslie
[1] www.who.int/health-topics/cancer#tab=tab_1
[2] www.cancerresearchuk.org/health-professional/cancer-statistics-for-the-uk#heading-Zero
[3] www.ncbi.nlm.nih.gov/pmc/articles/PMC6015544/
[4] www.cancerresearchuk.org/about-cancer?_gl=1*12i1zk4*_ga*MzU1ODQ3NTg0LjE2MzQ2NjYwNzQ.*_ga_58736Z2GNN*MTYzNTI1MTE4MS42LjEuMTYzNTI1MTIyNi4xNQ..&_ga=2.103174536.967622727.1635115294-355847584.1634666074
[5] www.en.wikipedia.org/wiki/Keratinocyte/
[6] www.sciencedaily.com/terms/parietal_cell.htm/
[7] www.cancerresearchuk.org/about-cancer/what-is-cancer/how-cancer-starts
[8] www.en.wikipedia.org/wiki/Spalax
[9] www.pubmed.ncbi.nlm.nih.gov/16000366/
[10] www.sciencing.com/minimum-oxygen-concentration-human-breathing-15546.html
[11] www.ourworldindata.org/life-expectancy
[12] www.genome.gov/genetics-glossary/Non-Coding-DNA
[13] www.nature.com/scitable/topicpage/transposons-or-jumping-genes-not-junk-dna-1211/
[14] www.nature.com/articles/s41586-021-03542-y?proof=t
[15] www.nature.com/articles/s41590-021-01027-8